When ‘Normal’ isn’t Optimal - The Early Signs of Blood Sugar Dysfunction
Blood Sugar, Prediabetes, and Type-2 Diabetes
What’s Actually Happening in the Body - and What You Can Do About It
Blood sugar is one of the most talked-about topics in health - and one of the most misunderstood.
Most people are told, “Your glucose looks fine,” or “Your A1c is normal,” and they move on. Meanwhile, the underlying physiology may already be shifting in a direction that, over time, can lead to prediabetes or type 2 diabetes.
The goal of this article is simple: to help you understand what’s actually happening in the body, how we measure it correctly, and what truly helps.
How Blood Sugar Regulation Is Supposed to Work
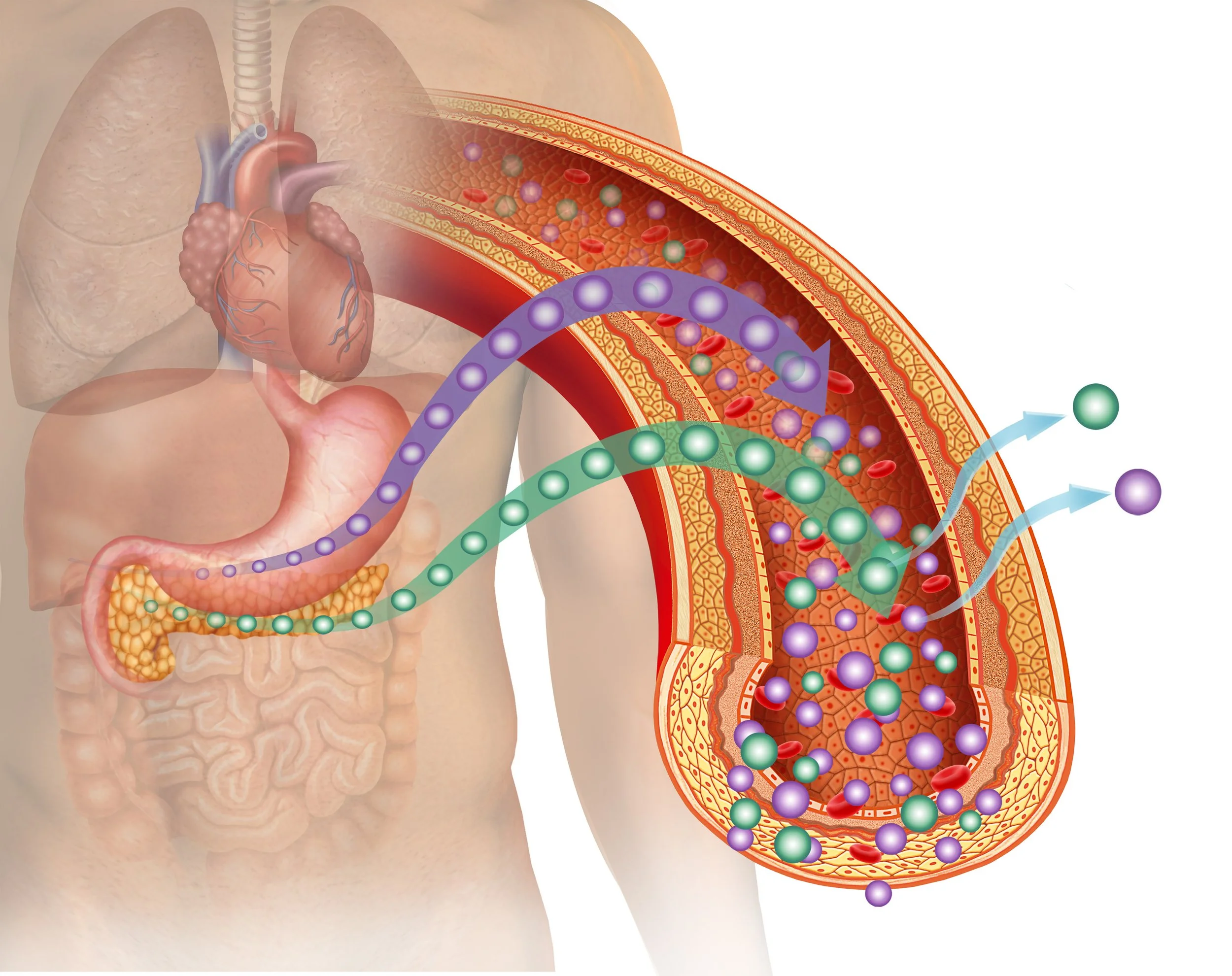
Pancreatic beta cells produce insulin, which is released into the bloodstream to help lower blood glucose levels.
When you eat, carbohydrates are broken down into glucose and released into the bloodstream.
In response, the pancreas releases insulin. Insulin allows glucose to move from the blood into the cells, where it is used for energy.
In a healthy system, this process is efficient and tightly regulated. Blood sugar rises after a meal, insulin responds appropriately, and levels return to baseline without much effort.
This is what metabolic health looks like.
What Changes in Prediabetes
Prediabetes is not a sudden condition - it is a gradual shift in how the body handles glucose.
The key issue is insulin resistance. This means the cells are no longer responding well to insulin. As a result, the pancreas compensates by producing more insulin to keep blood sugar in a normal range.
For a period of time, this works. Blood sugar may still look “normal,” but insulin levels are elevated behind the scenes.
Eventually, this compensation begins to fail. Blood sugar starts to rise, and prediabetes is diagnosed.
Clinically, this is often defined as:
HbA1c between 5.7–6.4%
Fasting glucose between 100–125 mg/dL
However, these markers often rise late in the process. Metabolic dysfunction is already well in place by the time prediabetes is diagnosed.
What Happens in Type 2 Diabetes
Type 2 diabetes develops when insulin resistance becomes more severe and the pancreas can no longer keep up with demand.
At this stage:
Blood sugar remains chronically elevated
Insulin may be very high, or it may begin to decline as pancreatic function deteriorates
Diagnostic criteria include:
HbA1c ≥ 6.5%
Fasting glucose ≥ 126 mg/dL
This is not just a “blood sugar problem.” It reflects a deeper disruption in metabolic signaling.
Why Elevated Blood Sugar Is Harmful
Chronically elevated glucose is not benign. It has direct effects on blood vessels, nerves, and organs.
One of the primary mechanisms is the formation of advanced glycation end products (AGEs). These occur when excess glucose binds to proteins and lipids in the body.
Over time, AGEs:
Damage the lining of blood vessels
Increase oxidative stress
Promote inflammation
This contributes to:
Atherosclerosis
Impaired circulation
Increased cardiovascular risk
High glucose also activates metabolic pathways that increase oxidative stress inside cells - especially in tissues like the eyes, kidneys, and nerves.
This is why prolonged hyperglycemia is associated with:
Neuropathy
Kidney damage
Retinal damage
In simple terms: extra glucose becomes chemically sticky and damaging over time.
The Missing Link: Stress, Cortisol, and Blood Sugar
This is the piece that many people - and frankly, many lab panels - miss.
When the body perceives stress (physical, emotional, or even just ongoing life pressure), it activates the hypothalamic-pituitary-adrenal (HPA) axis. This leads to the release of cortisol.
Cortisol is not a “bad” hormone. It’s actually essential for survival. Its job is to make sure you have enough energy to respond to a stressor.
One of the ways it does this is by increasing blood sugar.
Specifically, cortisol:
Signals the liver to produce more glucose
Stimulates gluconeogenesis (making new glucose from amino acids and other substrates)
Reduces the effectiveness of insulin in peripheral tissues
This is well-established in the research. Cortisol has been shown to directly increase hepatic glucose production by stimulating gluconeogenesis, and stress hormones - including cortisol - act on the liver to increase glucose output.
In the short term, this is helpful. If you’re running from a threat, you want more glucose available.
But in modern life, the “threat” is often:
Chronic psychological stress
Poor sleep
Overtraining
Ongoing inflammation
And the response doesn’t turn off.
Over time, chronic stress:
Keeps cortisol elevated
Continually signals the liver to produce glucose
Increases insulin resistance
Creates a pattern of persistently elevated blood sugar
In fact, chronic stress has been shown to increase both gluconeogenesis and glycogen breakdown, while reducing insulin sensitivity - creating a metabolic environment that promotes diabetes .
There is also evidence that higher cortisol levels are associated with higher blood sugar and impaired glucose control in humans .
Clinical translation: You can be eating well and still struggle with blood sugar if your nervous system is chronically dysregulated.
This is not a mindset issue. This is physiology.
The Most Useful Lab Markers (and What They Actually Tell Us)
Standard labs often miss early dysfunction. A more complete picture requires looking at multiple markers together.
HbA1c
This reflects average blood glucose over approximately three months. It is useful but can miss early insulin resistance and is influenced by red blood cell turnover.
Fasting Glucose
This is a snapshot in time and tends to rise later in the disease process.
Fasting Insulin
This helps identify how hard the body is working to maintain normal glucose. Elevated levels suggest insulin resistance.
However, insulin is rapidly cleared by the liver, which can make it less stable as a marker.
C-Peptide
C-peptide is released in equal amounts to insulin when the pancreas produces it, but it is not rapidly cleared by the liver.
Because of this, it provides a more stable and accurate reflection of endogenous insulin production.
GlycoMark (1,5-anhydroglucitol)
This marker reflects short-term glucose control and is particularly useful for detecting post-meal blood sugar spikes that may not be captured by HbA1c.
Nutrition: The Most Powerful Intervention
Dietary changes are one of the most effective ways to improve blood sugar regulation.
Research consistently supports:
Reducing refined carbohydrates and added sugars
Increasing protein intake to improve satiety and stabilize glucose
Increasing fiber, particularly soluble fiber, to slow glucose absorption
Including healthy fats to reduce post-meal glucose spikes
There is no single “perfect” diet, but patterns that reduce glucose variability and insulin demand tend to be most effective.
Movement: A Key Part of the Equation
Skeletal muscle plays a major role in glucose disposal.
Resistance training improves insulin sensitivity by increasing muscle mass and enhancing glucose uptake.
Even moderate activity, such as walking after meals, has been shown to reduce postprandial glucose levels.
This is one of the simplest and most effective interventions available.
Sleep and Stress
Sleep deprivation and chronic stress both impair glucose regulation.
Elevated cortisol levels increase blood sugar and contribute to insulin resistance.
This is not a “secondary” issue - it is a central driver for many people.
Supplements: What Has Evidence
Some supplements have been studied for their effects on blood sugar regulation.
Berberine
Berberine has been shown in randomized trials to improve fasting glucose and HbA1c. Its mechanisms include activation of AMPK, improved insulin sensitivity, and effects on gut microbiota.
Several studies have found its glucose-lowering effects to be comparable to metformin in certain populations.
Magnesium
Magnesium plays a role in insulin signaling, and deficiency is associated with insulin resistance.
Alpha-Lipoic Acid
Improves glucose uptake and reduces oxidative stress.
Inositol
Supports insulin signaling and metabolic function.
Supplements can be helpful, but they work best when layered onto strong foundational habits.
A Brief Note on GLP-1 Medications
GLP-1 receptor agonists, such as semaglutide, mimic a natural hormone involved in glucose regulation.
They:
Increase insulin secretion
Decrease glucagon
Slow gastric emptying
Reduce appetite
They can be very effective tools - but they do not replace the need to address the underlying metabolic environment.
Final Thoughts
Blood sugar regulation is not just about glucose - it is about how the entire system responds to food, stress, movement, and environment.
By the time glucose and HbA1c are elevated, the process has often been developing for years.
A more complete approach - using the right lab markers and focusing on foundational lifestyle interventions - allows for earlier detection and more meaningful change.
And in many cases, that’s where everything starts to turn around.